XL 100
ALL FEATURES OF A LARGE ONE
XL 100 – Small R&D Rotary Tablet Press
The XL 100 features an integrated electrical cabinet and a very simple and robust mechanical design. The fully self-contained and portable machine is the ideal tool for the product development. The XL 100 offers extreme accessibility to the compression zone (no corner columns), an exchangeable turret for maximum flexibility, and a combination of quick-disconnects and smooth surfaces that permit fast cleaning and changeover.
Small R&D
Rotary
Tablet Press


Scale-Up
Rotary R&D Press
Full Instrumentation Available
Exchangeable Turret

Designed for Pharma & Nutraceuticals
With a versatile design concept, the XL 100 R&D tablet press is suited either for product development, scale-up, or clinical batch production. The machine complies with the highest standard in GMP and all valid FDA regulations. KORSCH controls permit full compliance with 21 CFR Part 11.

Small Product Quantities
The XL 100 permits the execution of full compaction studies with limited material quantities. A reduced volume product hopper and reduced volume feeder insure efficient operation, and the control system is fully functional with reduced press tools. The mixed turret design permits the development of B and D size tablets on the same turret.

Performance
The machine offers a precompression capability of 10 kN and a main compression capability of 60 kN and a maximal output of 86,400 tablets/h with a maximum press speed of 120 RPM.
The 120 RPM press speed permits meaningful data on compression dwell time and feeder dwell time to be developed and analyzed.
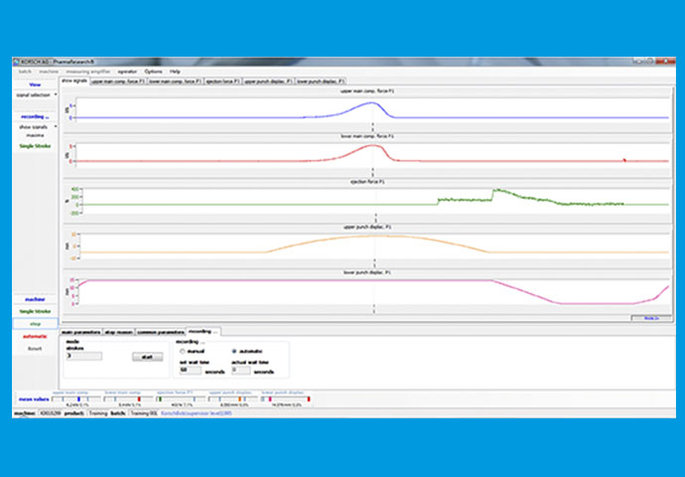
Instrumentation
The XL 100 may be fully instrumented for the measurement of precompression force, main compression force, ejection force, and scrape-off force, to permit product development parameters to be evaluated and stored.
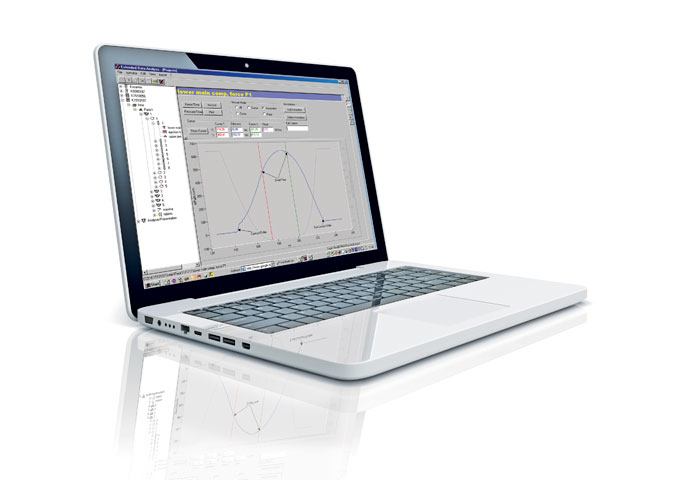
PharmaResearch
Together with the PharmaResearch, a Windows based data acquisition system and a full instrumentation package, the XL 100 permits storage, analysis, and export of compression and ejection force data.
PharmaResearch assists during:
- R&D and scale-up phase
- Production of small batches while monitoring limits
- Tableting process offering troubleshooting support
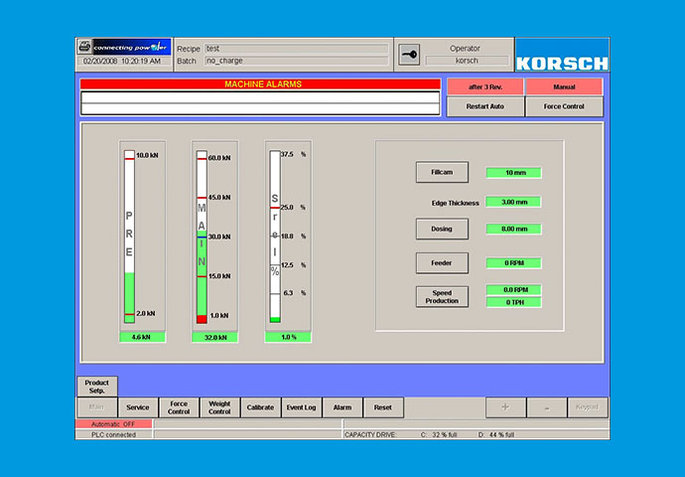
Optimal Control
As an option, the XL 100 offers the integrated press force control PharmaControl® to permit automatic weight control and single-tablet rejection.

Fully Portable
- Stainless steel base
- Locking casters
- Permits full portability
- Leveling footpads included

XL 100 Pro
Instead of a simple operator panel, the XL 100 Pro includes an industrial PC like the production machines. This allows to integrate the optional software PharmaResearch as well as the press force control PharmaControl® that permits additionally data recording. With a 15 inch touch screen, the XL 100 Pro control system offers a graphical and user-friendly environment.

Minimal Noise and Vibration
The unique and patented design of the carrier plate, with dampers, fully isolates vibration from the head piece and machine base. The result is:
- Very low noise emission < 80 dB(A)
- No vibration transmission to the floor of the compression room
- No segregation of powder in the feeding system which can occur with machine vibration
Containment Solutions
KORSCH offers a wide range of containment solutions on most machine models, including DryCon® and WipCon® executions. With proven solutions across the full containment range (OEB 3, OEB 4, OEB 5), KORSCH provides a fully turnkey system which is engineered to meet the specific requirements of the project. In partnership with the customer, KORSCH specialists define critical process parameters, including containment requirements, peripheral integration, and automation design. KORSCH engineers are expert in developing the concept, execution, integration, and final SMEPAC testing to fully validate the containment capability.


XL 100 WipCon®
The XL 100 WipCon® is an innovative, small-scale rotary tablet press that offers a comprehensive Wash-in-Place and high-containment capability for product development, scale-up, and clinical batch production of highly active or potent compounds. In concert with contained peripheral components, the XL 100 WipCon® offers a turnkey and proven R&D solution.
XL 100 Downloads
Download our brochure or have a look at the technical data:



