Smallest R&D Rotary Tablet Press
The smallest R&D rotary tablet press that offers all the features of larger tablet presses for single-layer applications. The XL 100 Tablet Press is a portable, robust, small scale rotary press and widely considered to be the standard for product development.

Turret Flexibility
The exchangeable turret permits the use of any size of standard press tools, including EU, TSM, B, D, and BB. The mixed turret design enables scientists to develop B and D size tablet formats on the same turret.

Ideal for Development, Scale-Up, and Clinical Batches
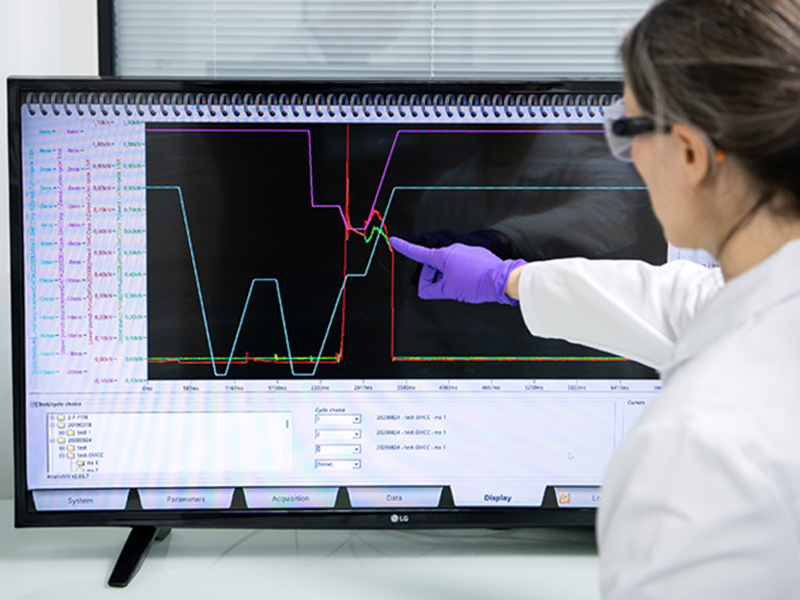
Optimal Controls and Instrumentation
The XL 100 may be fully instrumented to permit comprehensive data collection analysis with the KORSCH R&D software. An optional production control module permits the manufacture of clinical batches with press force control, single-tablet rejection, and full array of electronic audit trails.
Technical Details
Small R&D Rotary Tablet Press
| Single-Layer and Multi-Layer Capability |
Single-Layer
|
|---|---|
| Punch Stations | 13 / 12 / 10 / 8 |
| Compression Force | 10/60 kN |
| Tablet Diameter max. | 25 mm |
| Containment Capability | DryCon® and WipCon® |
| Tablet Output max. | up to 93,000 tablets/h |